Table 5
Gas phase chemical reactions involved in the destruction of gaseous ethanol (upper half of the table) and in the formation of gaseous OH, a major ethanol destroyer (lower half of the table).
| Reactants | Products | α (cm3/s) |
β | γ (K) |
Label | Ref. | |
|---|---|---|---|---|---|---|---|
| Ethanol destruction | |||||||
| C2H5OH + OH | → | CH3CHOH + H2O | 1.90×10−11 | 0 | 0 | 1a | Skouteris et al. (2018) |
| C2H5OH + OH | → | CH2CH2OH + H2O | 8.10×10−12 | 0 | 0 | 1b | Skouteris et al. (2018) |
| C2H5OH + H3O+ | → | H2O + C2H5OH+2 | 1.79×10−9 | −0.50 | 0 | 2 | KIDA, see text |
| OH formation | |||||||
| H2O + NH3+ | → | NH4+ + OH | 1.0 | 9.39×10−10 | 5.41 | 3 | KIDA & UMIST |
| CH3OH+2 + e− | → | H + CH3 + OH | 4.54×10−7 | −0.59 | 4 | UMIST | |
| H3O+ + e− | → | H + H + OH | 2.60×10−7 | −0.50 | 0 | 5 | UMIST |
Notes. The first two columns report the reactants and products, the following three columns the α, β and γ parameters. In reactions 1, 2 and 5, the rate constant k is computed by the usual Arrhenius-like equation: 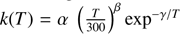 . In reaction 3, α represents the branching ratio of the channel and β the langevin rate. In reaction 4, the following holds:
. In reaction 3, α represents the branching ratio of the channel and β the langevin rate. In reaction 4, the following holds: 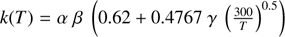 . Last column lists the references to the works and databases that report the reactions.
. Last column lists the references to the works and databases that report the reactions.
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.


