Fig. 5
Download original image
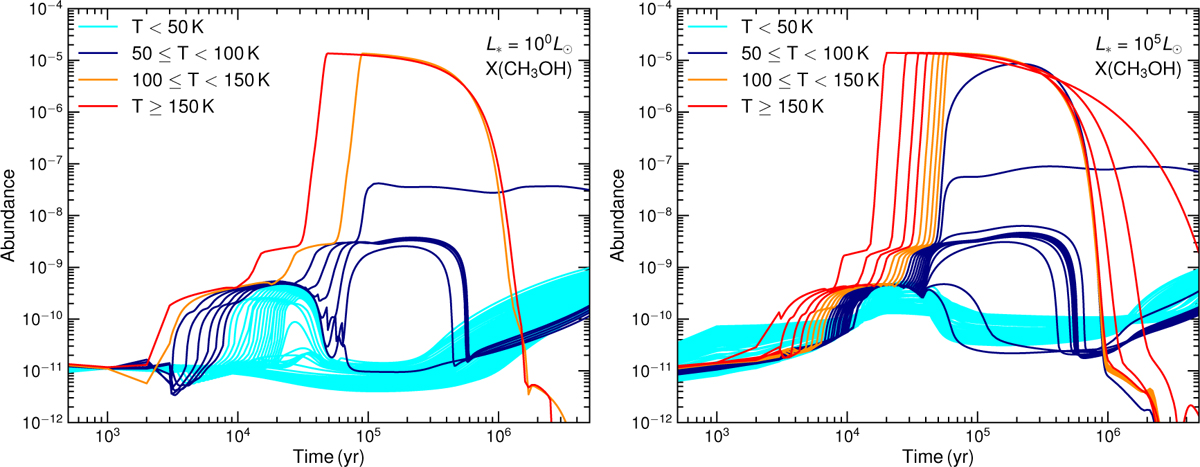
Example of the variation in CH3OH with time for L* = 100 L⊙ (left) and L* = 105 L⊙ (right). Lines are for the 100 parcels (full grid) from the center, color-coded by the temperature at t = 106 yr. The abundance of CH3OH decreases sharply at late times because of the reaction with neutral and ion species. For instance, at t = 5 × 105 yr, the most important destruction reactions are H3O+ + CH3OH → CH3OH2+ + H2O and H3+ + CH3OH → CH3+ + H2O + H2.
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.


