Table 2
Important reactions and their rate coefficients, taken from UMIST2012 database.
| Reaction no. | Reaction | Rate coefficient (cm3 s−1) |
|---|---|---|
| (R2) | 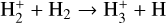 |
k2 = 2.08 × 10−9 |
| (R3) | 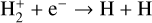 |
k3 = 1.6 × 10−8(T/300)−0.43 |
| (R4) | 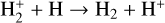 |
k4 = 6.4 × 10−10 |
| (R5) | 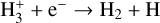 |
k5=2.34 × 10−8(T/300)−0.52 |
| (R6) | 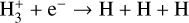 |
k6=4.36 × 10−8(T/300)−0.52 |
| (R7) | 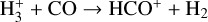 |
k7=1.36 × 10−9(T/300)−0.14 exp (3.4/T) |
| (R8) | 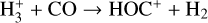 |
k8=8.49 × 10−10(T/300)0.07 exp (−5.2/T) |
| (R9) | 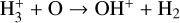 |
k9=7.98 × 10−10(T/300)−0.16 exp (−1.4/T) |
| (R10) | 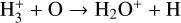 |
k10=3.42 × 10−10(T/300)−0.16 exp (−1.4/T) |
| (R11) | 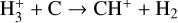 |
k11=2.00 × 10−9 |
| (R12) | HCO+ + e− → H+CO | k12=2.40 × 10−7(T/300)−0.69 |
| (R13) | 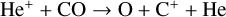 |
k13 = 1.60 × 10−9 |
| (R14) | He++O2 → O++O+He | k14 = 1.10 × 10−9 |
| (R15) | C++e− → C+h ν | k15 = 2.36 × 10−12(T/300)−0.29 exp (17.6/T) |
| (R16) | C++O2 → CO++O | k16 = 3.42 × 10−10 |
| (R17) | C++O2 → CO+O+ | k17 = 4.54 × 10−10 |
| (R18) | 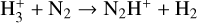 |
k18 = 1.80 × 10−9 |
| (R19) | CO + N2H+ → HCO++N2 | k19=8.80 × 10−10 |
Notes. Numerical subscripts on rate coefficients correspond to the reaction numbers stated in the text. The H3+–electron recombination rate coefficient (ke) is equal to k5+k6. The rate coefficient for proton transfer to CO, denoted as kCO, is calculated as k7+k8. The rate coefficient for destruction via proton transfer to O, kO, is equal to k9+k10. The rate coefficient for O2 destruction via reaction with C+, 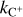 , is equal to k16+k17. Temperature-dependent coefficients are expressed in terms of gas kinetic temperature (T).
, is equal to k16+k17. Temperature-dependent coefficients are expressed in terms of gas kinetic temperature (T).
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.


