| Issue |
A&A
Volume 706, February 2026
|
|
|---|---|---|
| Article Number | A260 | |
| Number of page(s) | 16 | |
| Section | Planets, planetary systems, and small bodies | |
| DOI | https://doi.org/10.1051/0004-6361/202555595 | |
| Published online | 18 February 2026 | |
Development of a C/H/O/N/S chemical network: Experimental benchmark, application to exoplanets, and identification of key C/S coupling pathways
1
Université Paris Cité and Univ Paris Est Creteil, CNRS, LISA,
75013
Paris,
France
2
Université de Lorraine, CNRS, LRGP,
54000
Nancy,
France
3
Department of Physics and Astronomy, University College London,
Gower Street,
London
WC1E 6BT,
UK
4
Astrophysics Group, University of Exeter,
Exeter
EX4 4QL,
UK
★ Corresponding author: This email address is being protected from spambots. You need JavaScript enabled to view it.
Received:
20
May
2025
Accepted:
24
October
2025
Abstract
Context. The detection of SO2 in WASP-39 b and WASP-107 b has increased interest in modeling photochemistry in exoplanets, especially sulfur compounds, creating an urgent need for reliable kinetic networks. However, sulfur kinetics data remain scarce, and comprehensive networks in combustion literature are only recent for H/O/S, in progress for C/H/O/S, and almost nonexistent for the full C/H/O/N/S system. Current exoplanet networks usually neglect this coupling by adding a sulfur sub-mechanism to C/H/O/N networks.
Aims. We integrated sulfur kinetics into our previously developed C0-C2/H/O/N chemical network, including coupling with carbon and nitrogen chemistry, for conditions between 500–2500 K and 100–10−6 bar. The reliability of the resulting network was a major focus.
Methods. We combined available combustion networks with original ab initio calculations where data were lacking, and compared the network with 1606 experimental measurements from combustion and pyrolysis of H2S, CH3SH, CS2, and OCS. We then applied it to six exoplanets (GJ 436 b, GJ 1214 b, HD 189733 b, HD 209458 b, WASP-39 b, and WASP-107 b) using the 1D kinetic model FRECKLL, and computed transmission spectra with TauREx 3.1. Results were compared with other sulfur networks used in exoplanet modeling.
Results. The new network includes 226 species and 1692 mostly reversible reactions. Carbon–sulfur coupling strongly impacts abundance profiles and observables. We predict that the abundance of CS2 is much higher than in current networks, while CH2S emerges as a key species to describe this coupling.
Conclusions. The significant role of C–S species underscores the need for chemical networks to be extensively tested against experimental data to improve reliability, particularly in the JWST era. The detection of CS2 in TOI-270 d further shows the need to address the blind spots in current models. Combustion and pyrolysis data prove to be valuable tools for this task.
Key words: astrochemistry / planets and satellites: atmospheres / planets and satellites: composition / planets and satellites: gaseous planets / planets and satellites: individual: WASP-39 b / planets and satellites: individual: WASP-107 b
© The Authors 2026
 Open Access article, published by EDP Sciences, under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Open Access article, published by EDP Sciences, under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This article is published in open access under the Subscribe to Open model. This email address is being protected from spambots. You need JavaScript enabled to view it. to support open access publication.
1 Introduction
Over the past several decades, and continuing today, the chemical characterization of exoplanet atmospheres has relied on a sustained community effort. This difficult and delicate task has generated many research programs on the acquisition and treatment of observational data for these celestial bodies, but also on the modeling of these data.
The commissioning of the James Webb Space Telescope (JWST) in July 2022 has flooded the exoplanet community with unprecedented, high-quality data, enabling rapid progress in our understanding of these worlds.
Forthcoming observational programs, including space-based missions such as ARIEL, scheduled for launch in 2029 (Tinetti et al. 2018), and ground-based instruments such as the Extremely Large Telescope, scheduled for first light in 2028, will intensify the need for precise modeling to ensure that these data are used to their full potential. The present study is motivated by recent results acquired by the JWST, which reveal the presence of SO2 in WASP-39 b and WASP-107 b (Tsai et al. 2023; Dyrek et al. 2024; Powell et al. 2024; Welbanks et al. 2024).
The implications of these detections are twofold. Firstly, SO2 is the first sulfur-bearing species ever detected in an exoplanet atmosphere, which confirms the importance of sulfur chemistry for exoplanets, as it demonstrates that these species can affect the spectrum. This opens the door to its quantification and potentially to retrieving information on the planet’s formation through the ratios with other elements (Cridland et al. 2019).
Secondly, SO2 is thermodynamically unstable in these atmospheres, as it is a highly oxidized form of sulfur. In the hydrogen-dominated atmospheres of hot Jupiters such as WASP-39 b and WASP-107 b, the thermodynamically stable form of sulfur is H2S, which, unlike SO2, has not been unambiguously detected. This indicates that photochemical processes oxidize H2S to SO2 (Tsai et al. 2023), making it necessary to include these species in models to estimate their chemical abundances and understand their impact on synthetic spectra. This is generally neglected in most retrieval analyses, which typically assume thermochemical equilibrium or constant abundance profiles (Wakeford et al. 2017; Kreidberg et al. 2014). This is because this modeling is computationally intensive as it requires integrating the chemical kinetics occurring inside the atmosphere until the steady state is reached. These chemical processes are represented as a kinetic network, the size of which increases rapidly with its comprehensiveness and can become prohibitive for 3D modeling or retrievals. However, for these applications, this network size can be reduced using different reduction approaches (Lira-Barria et al. 2022; Venot et al. 2019; Tsai et al. 2022; Yang & Hu 2024) and used to perform retrievals with TauREx thanks to its new FRECKLL plugin (Al-Refaie et al. 2024). Another constraint on the modeling of chemical kinetics is its reliance on two types of crucial data.
The first type is accurate photolysis data, which are available for most major species at room temperature, but can deviate drastically under the high-temperature conditions of exoplanet atmospheres (Venot et al. 2013; Venot et al. 2018; Fleury et al. 2025). The second is kinetic data for the reactions in the network, whose rates are parameterized with rate constants that depend heavily on temperature and also pressure for some reaction types. While data for earth-like conditions (300 K, N2–O2 atmosphere) are available for simple and abundant species, higher-temperature data for hydrogen-dominated atmospheres are more difficult to gather. Several approaches have been adopted to address this problem. The most common approach is to gather multiple rate constants from various databases, such as KIDA (Wakelam et al. 2012) or the NIST chemical kinetics database (Manion et al. 2008). On the one hand, this approach can lead to small network sizes, which lower computational cost by enabling the inclusion of only relevant species. On the other hand, it is biased toward including only reactions available in the literature that directly involve the main species. This can lead to internal inconsistencies in the network and the neglect of crucial species and reactions to accurately model the kinetics of the species of interest.
To solve this issue, networks directly derived from those used in combustion modeling were developed, as these usually undergo extensive validations1 against experiments in ideal reactors (Venot et al. 2012; Venot et al. 2015; Venot et al. 2020). Our previous work also focused on deriving such a network from the most recent advances in combustion and pyrolysis kinetics for C/H/O/N compounds and separately performing validation against a wide range of combustion experiments (Veillet et al. 2024).
We take the same approach in this work, starting from available sulfur networks in the combustion and pyrolysis literature that are used to model the thermal decomposition of hetero-atomic organic compounds in many combustion applications. These compounds may contain oxygen, sulfur, nitrogen, phosphorus, or halogens, and are the building blocks of much combustion-relevant matter, such as materials (Xu & Hadjichristidis 2023), pesticides (Casida & Durkin 2017), toxic agents (Sirjean et al. 2017), pharmaceuticals, or fuels, either derived from organic matter or fossil resources, such as biogas or coal-derived syngas (Mathieu et al. 2015). The combustion of these hetero-atomic compounds is studied both for applications under controlled conditions, such as the incineration of waste and its energy recovery, but also to prevent accidental combustion such as fires and explosions. Forest fires also fall into this category, in which biomass containing hetero-atoms is burned. These phenomena involve both solids and gases, and, in most cases, the first step required for combustion to take place is an initial pyrolysis stage (Stauffer et al. 2007). For the combustion of gases with impurities, the literature shows that the presence of hetero-atoms such as sulfur or nitrogen perturbs the reactivity of organic compounds and the type and quantity of pollutants emitted (Mathieu et al. 2014; Honorien et al. 2023; Glarborg et al. 2018). In combustion processes, the rapid decomposition of fuel is followed by fragment oxidation, which is kinetically limiting in the fuel oxidation process (Wang et al. 2018). Robust reaction bases, which have been validated over a wide range of pressure and temperature conditions, are therefore also required in combustion research on hetero-atomic organic compounds, where the temperatures encountered are similar to those found in hot exoplanets. Although these core reaction mechanisms are widely available for hydrocarbon combustion, they remain scarce for describing the combustion chemistry of hetero-atomic compounds containing both sulfur and nitrogen. Nitrogen chemistry sub-mechanisms were originally included along with the combustion chemistry of small hydrocarbons for NOx formation. Recently, some of the nitrogen sub-mechanisms also account for NOx reduction reactions with ammonia (Glarborg et al. 2018). Kinetic models incorporating the combustion chemistry of sulfur compounds were developed mainly for H2S, to account for its presence as an impurity in some types of gas and, to a lesser extent, for CH3SH (Stagni et al. 2022; Mathieu et al. 2014; Zhou et al. 2013; Cooper et al. 2022). In this work, we used experimental and theoretical combustion and pyrolysis data, along with our previous work on a C/H/O/N chemical network (Veillet et al. 2024) and CH3SH kinetics (Veillet et al. in rev), to develop our network for sulfur modeling in exoplanets, with fully coupled C/H/O/N/S interactions, especially between carbon and sulfur. This C/H/O/N kinetic network has been statistically validated against a database of 1618 experimental combustion and pyrolysis datasets (Veillet et al. 2024), featuring CH4, CH3OH, C2H5OH, CO, H2, H2O, NO, HCN, NH3 and their mixtures. We used a similar approach to select a H2S kinetic model from the literature, based on extensive comparisons with a newly developed sulfur combustion database (SOx, H2S, and CS2) composed of 1151 experimental measurements. For methanethiol (CH3SH), the main link in the carbon-sulfur chemistry, we propose a new detailed pyrolysis and combustion mechanism based on previous theoretical calculations and validate it against recent experimental data (Colom-Díaz et al. 2021; Alzueta et al. 2019). We then used this kinetic network to model both hot Jupiters and warm Neptunes, and to investigate the effect of sulfur chemistry on other species and their synthetic transit spectra.
Section 2 discusses the choices and available data used to build the C/H/O/N/S kinetic network, both theoretical and experimental. In Sect. 3, this network is used to model the atmospheres of six exoplanets: GJ 436 b, GJ 1214 b, HD 189733 b, HD 209458 b, WASP-39 b, and WASP-107 b. We also perform a detailed chemical analysis on the main species of interest to identify the main contributing reactions and formation pathways for these species, and to investigate how the coupling between sulfur, carbon, and nitrogen chemistry impacts their abundances. The corresponding synthetic spectra are then simulated to evaluate the impacts on observables. Finally, we conclude in Sect. 4 and discuss potential future improvements to this work.
All sulfur experimental data used for mechanism validation.
2 Detailed combustion network selection
2.1 Considered combustion networks
The starting point of this work is a recently developed C0–C2 C/H/O/N core mechanism, built based on a systematic comparison of the performance of several available mechanisms from the literature for C/H/O and C/H/O/N chemistry against experimental data (Veillet et al. 2024). The kinetic model includes oxygenated or nitrogen compounds such as methanol, ethanol, ammonia and hydrogen cyanide, and small hydrocarbons (C2H6, C2H4, C2H2, and CH4). We gathered experimental data on combustion and pyrolysis of these species from the literature, covering a wide range of temperatures (800–2400 K) and pressures (0.2–50 bar). The experimental measurements included in this dataset consist of ignition delay times and species profiles as a function of temperature, time, and pressure in shock tubes, plug flow, jet-stirred, and closed reactors. To develop the present C/H/O/N/S mechanism, we built on this previous work and added sulfur chemistry following the same methodology. We gathered experimental measurements of the combustion and pyrolysis of four different species: H2S, CH3SH, OCS, and CS2. This dataset consisted of species profiles as a function of temperature in flow reactors and jet-stirred reactors. The resulting dataset, composed of 1606 experimental measurements, is summarized in Table 1. We then compared four kinetic models from the literature on this dataset: Stagni et al. (2022), Cooper et al. (2022), Zeng et al. (2021), and Colom-Díaz et al. (2021), which we briefly describe below.
Stagni 2022: a mechanism for H2S combustion and pyrolysis from Stagni et al. (2022), built and validated on a large review of H2S experimental data. It is one of the state-of-the-art model for H2S combustion and pyrolysis kinetics. No carbon chemistry is included.
Colom-Diaz 2021: a mechanism for CH3SH combustion and pyrolysis from Colom-Díaz et al. (2021), which iterates on previous work on atmospheric CH3SH kinetics.
Cooper 2022: a state-of-the art mechanism for H2S, built on the work of Haynes and co-workers and Glarborg and coworkers, which includes its oxidation by N2O from Cooper et al. (2022). This mechanism is one of the few models that attempt to describe N/S coupling.
Zeng 2019: a mechanism for CS2 combustion from Zeng et al. (2019), based on the CS2 kinetics from a previous mechanism proposed in Glarborg et al. (2014).
2.2 Comparison with experiments
To test the accuracy of these models on H/O/S chemistry, we first compared them with our H2S combustion and pyrolysis data using the Cantera software (Goodwin et al. 2023). Figs. 1 and 2 show two examples of these comparisons. In these comparisons, the model from Stagni et al. (2022) shows the best agreement with the experimental data. This is especially true for H2S pyrolysis, since the other three models show an offset of approximately 100 K in their H2S consumption and H2 production. For the combustion data, the Cooper 2022 model also performs well, with a temperature shift of around 50 K in H2S consumption and SO2 production. By contrast, the Zeng 2019 and Colom-Diaz 2021 models are in poorer agreement with the experimental data. The Zeng 2019 model shows a temperature shift of around 150 K, while the Colom–Diaz model exhibits strong variations (likely due to numerical instabilities) in H2S consumption and SO2 production between 700 and 850 K. We observe these variations in all simulations of the H2S combustion dataset for this model with different convergence parameters; therefore, they are not due to convergence issues.
Consequently, to implement H2S chemistry in the present work, we merged the Stagni 2022 model with our previous C/H/O/N mechanism (Veillet et al. 2024). As Stagni 2022 already contains an H/O reaction subset, we replaced it with the one from our previous study and verified that this did not affect its performances on our H2S experimental dataset. Because the Stagni 2022 model only described H/O/S chemistry, C/S coupling had to be added to create our final mechanism. To fully cover the C/H/O/S chemistry, we first included the Colom-Diaz 2021 and Zeng 2019 models in our mechanism, excluding their H2S chemistry subset, which was already included from Stagni 2022. As the Colom-Diaz 2021 model mainly described CH3SH combustion and pyrolysis, it also contains a CS2 reaction subset, which we replaced with the one from Zeng 2019, which specializes in CS2 and OCS combustion. However, as shown in Fig. 1, the Colom-Diaz 2021 model exhibits numerical issues, which led us to develop a new kinetic model for CH3SH. Most parts of this new sub-mechanism were estimated by analogies with close analog oxygenated species sharing the same molecular geometry (i.e., CH3OH for CH3SH, CH2O for CH2S). Activation energies were corrected for differences in reaction enthalpy when necessary, and a total of more than 120 reactions were included in this CH3SH. The rate coefficients of the most sensitive reactions for pyrolysis simulations and unknown thermochemistry data were calculated using ab initio calculations at the CCSD(T)-F12/CBS level. Finally, to account for the N/S coupling, we added the corresponding reaction subset from Cooper 2022 to the present work, which included 40 additional reactions. This work is fully detailed in Veillet et al. (in rev) and is not described in more detail here.
 |
Fig. 1 Comparison between models from the literature for the combustion of 500 ppm of H2S at atmospheric pressure and an air-to-fuel equivalence ratio of four in a flow reactor (no. 1 in Table 1). |
2.3 Thermal decomposition of H2S
The current understanding of the thermal decomposition of H2S remains debated and has required us to make important choices that we detail here. The literature review of Raj et al. (2020) on sulfur combustion, particularly H2S kinetics, further details the conflicts between modeling and experiment, also noted in Stagni et al. (2022). In essence, the two reactions in question are the possible pathways for thermal decomposition of H2S: H2S → SH + H and H2S → S + H2. The first pathway was the most implemented in early kinetic networks, including H2S pyrolysis (Higashihara et al. 1976; Bowman & Dodge 1977; Roth et al. 1982), as it is a simple bond-breaking reaction and a standard, well-known type of initiation reaction. The second pathway is a spin-forbidden reaction, asH2S and S are triplets and H2 a singlet. Consequently, this reaction must proceed through an intersystem crossing from the singlet potential energy surface to the triplet energy surface. Because this process is typically slow and difficult to model theoretically, it was initially ignored in models until experimental evidence for this decomposition channel was reported by Woiki & Roth (1994) and Olschewski et al. (1994). A subsequent theoretical study by Shiina et al. (1996) analyzed these potential energy surfaces and the crossing point energy, but the derived rate constant was an order of magnitude lower than those measured by Woiki & Roth (1994) and Olschewski et al. (1994). Furthermore, additional experimental measurements by Shiina et al. (1998) and Karan et al. (1999) confirmed the previous experimental values, indicating that this theoretical value was likely underestimated. Shiina et al. (1998) also experimentally measured the corresponding reverse reaction, H2 + S → H2S and compared it to the channel H2 + S → SH + H, showing that the latter is favored at 100 bar at 900 K. However, these experimental measurements are also subject to caution, as Zhou (2009) demonstrated that even quartz surfaces can catalyze H2S decomposition. Nonetheless, the activation energy deduced for H2S → S + H2 from these independent experiments is in direct conflict with ab initio calculations and thermochemical data, because the corresponding activation energy is lower than the reaction enthalpy. This results in a negative activation energy for the reverse reaction H2 + S → H2S, which favors this pathway over H2 + S → SH + H reaction, in direct conflict with the measurements of Shiina et al. (1998). Such an activation energy is unphysical, but it is the only one that reproduces both high- and low-temperature experimental data. To reconcile this activation energy with the correct reverse pathway, we used an intermediate species corresponding to the first excited state of the sulfur atom S(1D). This species was included in our mechanism by splitting the reaction H2S → S + H2 into two reactions: H2S → S(1D) + H2 and S(1D) S. The former was written with the same parameters as H2S → S + H2, while the latter used the same parameters as the analog reaction O(1D) → O(3P). For S(1D) thermochemical data, we used the formation enthalpy of the S(3P) ground state (denoted simply as S in this work) and added the energy difference with S(1D). For the entropy, we used the Sackur–Tetrode equation (Sackur 1913; Tetrode 1912). Because the S(1D) → S reaction is much faster than H2S → S(1D) + H2, the total reaction rate for the forward reaction remains unchanged. The reverse reaction is prevented by the step S → S(1D), which is very slow due to the high energy barrier. This approach effectively reconciles the observed activation energy with the preferred reverse pathway, although it does not resolve the issue of the non-physical activation energy for H2S → S(1D) + H2. Further theoretical and experimental studies on H2S pyrolysis are required to fully resolve this conflict, but this solution provides the best compromise given the current state of the literature.
 |
Fig. 2 Comparison between models from the literature for H2S pyrolysis (500 ppm at atmospheric pressure in a flow reactor, no. 2 in Table 1). |
Exoplanets simulated in this study and the input parameters.
3 Application to exoplanetary atmospheres
3.1 Models and data sources
Throughout the development of this experimentally validated C/H/O/N/S kinetic network, we focused on the coupling of sulfur chemistry with the rest of the network. We primarily aimed to model the impact of sulfur chemistry in hot exoplanets, both on sulfur species and on the main C/H/O/N species (such as H2O, CH4, CO, CO2, NH3, HCN, C2H2, andC2H4). In this section, we use this kinetic network to model the atmosphere of six exoplanets: three warm Neptunes (GJ 436 b, GJ 1214 b, WASP-107b) and three hot Jupiters (HD 189733 b, HD 209458 b, WASP-39 b).
WASP-39 b and WASP-107 b were chosen specifically because of recent observations of SO2 in their transit spectra by the JWST (Tsai et al. 2023; Dyrek et al. 2024), while HD 189733 b, HD 209458 b, GJ 436 b, and GJ 1214 b are planets that have been extensively studied and are commonly used to represent their respective exoplanet classes (Venot et al. 2012, 2020; Moses et al. 2011). The parameters used to model these planets (pressure-temperature profile, UV stellar flux, eddy diffusion coefficient, and metallicity) are the same as in Veillet et al. (2024) for GJ 436 b, GJ 1214 b, HD 189733 b, and HD 209458 b, while for WASP-39 b and WASP-107 b we used the same input parameters as Tsai et al. (2023) and Dyrek et al. (2024).respectively. Table 2 summarizes these parameters. We used the 1D model FRECKLL (Al-Refaie et al. 2024) to run the simulations with both our newly developed kinetic network and the sulfur kinetic network from VULCAN used in Tsai et al. (2023) to simulate WASP-39 b, hereafter referred to as V25 and Tsai 2023. The pressure-temperature profile was discretized in 130 layers, and the photodissociation data used for the sulfur species are listed in Table A.1. Fig. 3 shows the obtained abundance profiles. To study the effect of the coupling on the C/H/O/N species, we also compared our results with those obtained using the scheme developed in Veillet et al. (2024) (without sulfur but featuring the same C/H/O/N chemistry), hereafter referred to as V23, is shown in Fig. 4. The corresponding synthetic transmission spectra at a resolution of 50 were also generated using TauREx (Al-Refaie et al. 2021). We used opacity data from Exomol (Tennyson & Yurchenko 2012) for H2O, CH4, CO, CO2, NH3, HCN, C2H2, C2H4, H2CO, OH, CH3, CN, NS, SH, SO, SO2, SO3, H2S, CS, and OCS, and HITRAN (Gordon et al. 2022), downloaded from the petitRADTRANS website (Mollière et al. 2019), for CS2. Collision-induced absorption was included for H2 H2 and H2 He, using data from HITRAN (Gordon et al. 2022). The calculations also included the default Rayleigh scattering from TauREx. The resulting spectra are shown in Fig. 5.
In the following, we discuss the main differences observed in the synthetic spectra. Detailed contributions are shown in Fig. B.1. We investigated the causes of these differences using the abundance profiles. We computed the formation pathways responsible for changes in the abundance profiles by solving the minimum flow problem from graph theory using the Python package NetworkX. Nodes were chosen as species in the network while edges were determined from reactions, with a capacity equal to their rate. We determined the nodes linked by an edge for each reaction by maximum matching on a bipartite graph formed from the set of reactants and the set of products. We based the maximum match chosen for each reaction on a greedy algorithm (Chvatal 1979) using a cost matrix calculated from the number of identical and different atoms for each match. This choice favors links between the product that most closely resembles one of the reactants, with a bias toward reactants that share the highest number of identical atoms. This is optimal for simple abstraction bimolecular reactions, for example, but is limited for more complex cases such as association reactions. For this reason, when identifying pathways between species, we allow the user to specify which atoms must be included in all intermediate species. Therefore, when generating the graph to calculate the total flow, we only include species that contain these atoms and exclude those that did not fulfill this condition. This results in a directional graph that represents the chemical pathways available from one species to another, together with the corresponding flows. This approach enables quantitative characterization of both primary and secondary formation pathways. In the following, a chemical pathway (with its corresponding flow) refers to the list of intermediate species from the source to the sink. Each link between intermediate species is composed of the sum of the rates of all the reactions that link these species. Two important properties of these “steps” in the chemical pathway are therefore the contribution of each reaction to the total capacity of the link and the ratio of this capacity to the global flow between the source and sink, which we call saturation in the following. This saturation provides information on the sensitivity of the total flow of the chemical pathway to the reaction rates and is by definition 100% for the limiting step in the pathway. The global flow of this pathway will therefore be less sensitive to variations in the rates of reactions with low saturation.
 |
Fig. 3 Abundance profiles of GJ 436 b, GJ 1214 b, HD 189733 b, HD 209458 b, WASP-39 b, and WASP-107 b simulated with FRECKLL. Solid lines show results from the present work, while dashed lines show those from Tsai 2023. We omit H2 and He for clarity to focus on the main species. |
 |
Fig. 4 Abundance profiles of GJ 436 b, GJ 1214 b, HD 189733 b, HD 209458 b, WASP-39 b, and WASP-107 b simulated with FRECKLL. Solid lines show results from the present work, while dashed lines show results from V23. |
3.2 Results for GJ 436 b and GJ 1214 b
For GJ 436 b, we modeled the planet using a 10× solar metallicity (from Lodders (2010)), as exact values are uncertain but likely at least one order of magnitude above solar (Madhusudhan & Seager 2011; Moses et al. 2013). Recent data from the JWST (Mukherjee et al. 2025b) suggest that its metallicity could be closer to 100× solar, indicating that this value might be underestimated. In general, some planet parameters, such as metallicity, may deviate from current estimates or be revised by future data. When no exact literature value is available, we used general estimates that represent one of the possible cases. For GJ 1214 b, we chose a higher metallicity of 100× solar, as suggested by recent observations of JWST (Nixon et al. 2024; Kempton et al. 2023). For GJ 436 b, a constant eddy diffusion coefficient of 108 cm2/s was used in the absence of a precise profile from a General Circulation Model (GCM) calculations, while for GJ 1214 b, we adopted the same profile as in Veillet et al. (2024). The resulting abundance profiles, shown in Fig. 3, reveal several differences between our sulfur network (solid lines) and Tsai 2023 (dashed lines). Firstly, with our network, the quenching point of CO is slightly higher in the atmosphere of GJ 436 b, resulting in increased CO and CO2 above 10 bar. The sole pathway converting CO to CO2 is the single elementary reaction CO + OH → CO2 + H. Secondly, the abundance of CS2 is clearly higher with our network, especially between 1 and 10−4 bar, with a difference of two to three orders of magnitude for GJ 436 b and one to two orders of magnitude for GJ 1214 b. This discrepancy arises from differences in the CS2 formation mechanisms between the two networks. For V25, CS2 forms from CH4 through CH3, CH2S, CHS, and CS via the following steps:
 (1)
(1)
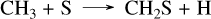 (2)
(2)
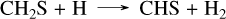 (3)
(3)
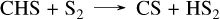 (4)
(4)
 (5)
(5)
This pathway represents 99.4% of the flow from CH4 to CS2, resulting in around 3.8 × 105 molecule cm−3s−1. The flows between each intermediate species involved in these steps are greater than 80% attributable to these reactions, except for the second, which contributes equally with the reaction CH3 + S2 → CH2S + SH to the formation of CH2S. The sulfur atom and disulfur species originate from the decomposition of the thermodynamically stable species H2S through the reactions H2S + H → SH + H2, SH + H → S + H2, and CS2 + S → CS + S2 as CS2 becomes abundant. The limiting step is CHS + S2 → CS + HS2, but the second and third reactions in this pathway, CH3 + S → CH2S + H or CH3 + S2 → CH2S + SH and CH2S + H → CHS + H2, are also very close to saturation (i.e., they are rate-limiting, with 99.2 and 92.3%, respectively). The primary sink of CS2 is its advection to lower atmospheric layers and destruction via oxidation to OCS and subsequently CO through the steps CS2 + S → CS + S2, CS + SO → OCS + S, and OCS + H → CO + SH, with the second reaction as the limiting step. This formation pathway is absent from the Tsai 2023 network because CH2S is not included. This results in a very reduced flow of 1.4 × 10−6 molecule cm−3s−1, which is equivalent to the flows from other carbon-bearing species such as CO, CO2, HCN, and OCS. We are confident that this pathway is accurate because it arises through reactions that were investigated in our preliminary study on CH3SH pyrolysis (Veillet et al. in rev). The corresponding experimental data (no. 3 in Table 1), shown in Fig. D.1, were compared to the ideal reactor simulations with the present work and Tsai 2023. These conditions clearly show that the Tsai 2023 kinetic network lacks a formation mechanism to explain the experimental CS2 abundance observed during the pyrolysis of CH3SH. In particular, the formation pathway and sensitivity analysis in this study demonstrate that CH2S is the key species that controls the formation of CS2. Precise ab initio calculations have been performed on this species, and the reaction CH3 + S2 → CH2S + SH is calculated in Veillet et al. (in rev) with full pressure dependence by solving the master equation for the full potential energy surface. However, many analogies were also used in this work given the sheer number of possible reactions, and further thorough studies are required to fully describe the kinetics, particularly under oxidation conditions. This will be the focus of a follow-up study aiming to improve this oxidation mechanism (Veillet et al., in prep). This species has already been observed as a product of photochemistry experiments (He et al. 2020), and the detection of CS2 in TOI-270 d (Holmberg & Madhusudhan 2024; Benneke et al. 2024) further supports the likelihood that high concentrations of CS2 are induced by photochemistry. In addition, CS2 has been highlighted in the exoplanet photochemistry modeling literature (Mukherjee et al. 2025a; Zahnle et al. 2009), particularly for cooler exoplanets, a trend also observed in the planets studied here. Recent work (Moses et al. 2024) also highlights the same formation pathway for CS2, but notes that OCS chemistry, which is closely linked to CS2 oxidation, remains uncertain. This suggests that greater effort is needed to validate networks against the widest possible range of available data to highlight these blind spots in our models.
Fig. 4 shows the effect of coupling sulfur chemistry to C/H/O/N species by comparing the present sulfur network (solid lines) with the previous C/H/O/N network, V23 (dashed lines). For highly abundant species such as H2O, CH4, CO, andCO2, we observe very few consequences. For C2H2 and C2H4, however, we observe a higher production when including sulfur species in the network. At 10−3 bar, for example, C2H2 is more than two orders of magnitude more abundant in GJ 436 b, and C2H4 is between one and two orders of magnitude more abundant when using the new sulfur network. For GJ 1214 b, the magnitude impact on C2H4 is similar, whereas for C2H2 the effect is less than one order of magnitude. In the absence of sulfur, the normal formation pathway for C2H4 from CH4 is as follows:
 (6)
(6)
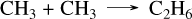 (7)
(7)
 (8)
(8)
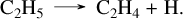 (9)
(9)
However, in the presence of sulfur, this pathway accounts for only 2.5% of the C2H4 produced. Most of the production skips the second step CH3 + CH3 →C2H6, which is the limiting reaction, and proceeds directly to C2H4 through the reaction CH2S + CH3 → C2H4 + SH. This reaction was also computed in our CH3SH pyrolysis study (Veillet et al. in rev), with the full pressure dependence calculated using the same master equation method. This result highlights the benefits of cross-validation between exoplanet modeling and combustion or pyrolysis experiments. The pathway to C2H2 with sulfur involves the reaction CH2S + CHS → C2H2 + HS2, which was an analogy with the equivalent addition CH2O + HCO → CH2OCHO. As the accuracy of this analogy is uncertain, further studies are required to confirm or disprove the reality of this reaction.
These abundance differences have very little impact on the synthetic transmission spectrum of GJ 436 b between 1 and 20 µm. However, around 25 µm, a new and significant CS2 feature of approximately 200 ppm is visible. Fig. B.1 further highlights the CS2 contribution to the spectra, showing that the features around 6.5 µm and 11 µm are also due to CS2, although they remain too small to significantly outweigh the NH3 features. For GJ 1214 b, abundance changes due to the chemical scheme have a more significant impact on the spectrum, although much of the spectral differences are due to an increase in the continuum, probably due to the large fraction of heavy elements in the atmosphere. The CS2 feature at 25 µm is also present, and a new CS2 feature at 11 µm can be seen next to the C2H4 feature.
 |
Fig. 5 Synthetic transmission spectra of GJ 436 b, GJ 1214 b, HD 189733 b, HD 209458 b, WASP-39 b, and WASP-107 b simulated with TauREx at a resolution of 50 for the abundance profiles shown in Figs. 3 and 4. |
3.3 Results for HD 189733 b and HD 209458 b
To model HD 189733 b and HD 209458 b, we adopted solar metallicity, as this appears to be a reasonable approximation based on known values (Xue et al. 2024; Finnerty et al. 2024). We also used the same eddy diffusion coefficient profiles as in Veillet et al. (2024). The corresponding abundance profiles of Fig. 3 show similar differences to those observed for GJ 436 b and GJ 1214 b in Sect. 3.2. CS2 is more abundant with our sulfur kinetic network than with Tsai 2023 by one to two orders of magnitude, due to the formation pathway not present in Tsai 2023. However, while this species reaches more than 1 ppm in HD 189733 b, its abundance remains low in HD 209458 b, peaking at 10−9 between 10−3 and 10−2 bars. Another notable difference is the CH4 abundance in HD 189733 b, which is lower in our network than in Tsai 2023. This is because in this layer of the atmosphere, the equilibrium between CS and CS2 is shifted toward CS. This causes a decrease in the CH4 abundance around 10−4 bar, which is not seen in the Tsai 2023 network due to the low abundances of these species. Other differences among the major species are mostly minor, although in the upper atmosphere of HD 209458 b H2S, CO2, and H2O abundances are slightly higher with our sulfur network than in Tsai 2023. Fig. 4 compares the abundance profiles obtained with our new sulfur scheme with those obtained with our network without sulfur. We observe several differences for HD 209458 b, mainly in NH3, CH4, C2H2, and HCN. However, these differences remain smaller than one order of magnitude. In contrast, for HD 189733 b, multiple C/H/O/N species are affected by the coupling with sulfur chemistry. The CH4 difference has already been discussed and is linked to the abundance of CS at this altitude. However, this directly impacts the HCN chemistry, which is formed from CH4 by the sequence of reactions:
 (10)
(10)
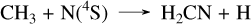 (11)
(11)
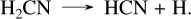 (12)
(12)
Remarkably, the reaction CH3 + N(4S) → H2CN + H resembles the previous reaction CH3 + S → CH2S + H. However, since the product is a radical, unlike in the CH2S case, it rapidly undergoes a β-scission to HCN. This close coupling between CH4 and HCN reduces the HCN abundance by approximately two orders of magnitude when accounting for sulfur, due to its impact on the CH4 abundance. The differences in the NH3 abundance profile are directly linked to this formation of HCN, as it also requires the nitrogen atoms that are primarily derived from NH3. At 10−4 bar in HD 189733 b, another notable coupling effect of sulfur chemistry is the main pathway of HCN formation from ammonia, which proceeds through NS, SNO, NO, HCNO, and HCN as follows:
 (13)
(13)
 (14)
(14)
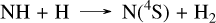 (15)
(15)
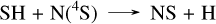 (16)
(16)
 (17)
(17)
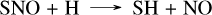 (18)
(18)
 (19)
(19)
 (20)
(20)
The oxidation of N(4S) to NO can also occur through via the reaction N(4S) + SO → NO + S. In the Tsai 2023 network, N(4S) reacts with CS to form HCN via two steps: N(4S) + CS → S + CN and H2 + CN → H + HCN. Although the effect is small, these reactions provide a clear example of the coupling between sulfur and nitrogen chemistry. The abundance profiles C2H2 and C2H4 are also affected by sulfur in two ways, due to the new formation pathway discussed in Sect. 3.2. Sulfur increases the abundances of these species near 10−2 bar, while the reduced CH4 abundance decreases them, dominating above 10−4 bar. For pressures lower than this, sulfur is mostly in the atomic form on both planets; however, there is a narrow pressure range around 10−3 bar where S2 dominates for HD 189733 b, while SH radicals dominate for HD 209733 b.
Looking at the synthetic transmission spectra of HD 209458 b and HD 189733 b (Fig. 5), we see that for HD 209458 b there is no significant change, except for slight deviations, particularly visible in the continuum above 10 µm. This can be understood from the spectra contributions (Fig. B.1), as the spectrum of HD 209458 b is entirely dominated by water. For HD 189733 b, however, several differences are notable. First, the CS2 feature at 25 µm is visible in the spectrum obtained with the abundances from our sulfur kinetic network. The second CS2 feature at 11 µm is also visible, with an amplitude of approximately 100 ppm. Second, the HCN feature at 13 µm disappears from the spectrum with our network compared to the Tsai 2023 network. Third, two amplitude changes occur at 3 and 7 µm, respectively. These correspond to CH4 features, which show higher amplitudes in the Tsai 2023 and V23 networks. This difference is due to the lower CH4 abundance obtained with our C/H/O/N/S network due to carbon-sulfur coupling, which causes fainter CH4 features.
3.4 Results for WASP-39 b and WASP-107 b
In order to stay as close to the original simulations as possible, we chose to model WASP-39 b and WASP-107 b with 10× solar metallicities, the eddy diffusion profile used in Tsai et al. (2023) for WASP-39 b, and a constant eddy coefficient of 1010 cm 2/s for WASP-107 b, as in (Dyrek et al. 2024). These two planets are the first in which the JWST has detected a sulfur species, namely SO2. This raises the question of how the differences between our network and Tsai 2023 impact the SO2 abundances and the corresponding synthetic spectra. For WASP-39 b, the abundance profiles in Fig. 3 show that the SO2 abundance profiles are very similar. This is because, in both kinetic networks, the formation mechanism of SO2 is the same,
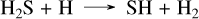 (21)
(21)
 (22)
(22)
 (23)
(23)
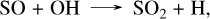 (24)
with the limiting step being the last one. This corresponds to the mechanism presented in Tsai et al. (2023), although the reaction rates are not exactly from the same source, which causes the observed differences. This high concentration of SO2 arises from the dissociation of H2S, which mainly forms S2 between 10−3 and 10−4 bar, and sulfur atoms below 10−4 bar. SO2 is not the only species that differs between the networks, we also observe that the CS2 abundance is higher in the present network than in Tsai 2023, as previously explained for all planets. This high concentration of sulfur atoms also impacts both NH3 and CH4. Indeed, N(4S) atoms are produced via the same mechanism as described when discussing the formation of HCN in Sect. 3.3, and they can then be oxidized by SH radicals through SH + N(4S) → NS + H. This reaction accounts for the differences in NH3 between the present sulfur network and Tsai 2023 seen around 10−4 bar for WASP-39 b in Fig. 4. For CH4, the abundance of SO oxidizes the carbon to CO2 via the following mechanism:
(24)
with the limiting step being the last one. This corresponds to the mechanism presented in Tsai et al. (2023), although the reaction rates are not exactly from the same source, which causes the observed differences. This high concentration of SO2 arises from the dissociation of H2S, which mainly forms S2 between 10−3 and 10−4 bar, and sulfur atoms below 10−4 bar. SO2 is not the only species that differs between the networks, we also observe that the CS2 abundance is higher in the present network than in Tsai 2023, as previously explained for all planets. This high concentration of sulfur atoms also impacts both NH3 and CH4. Indeed, N(4S) atoms are produced via the same mechanism as described when discussing the formation of HCN in Sect. 3.3, and they can then be oxidized by SH radicals through SH + N(4S) → NS + H. This reaction accounts for the differences in NH3 between the present sulfur network and Tsai 2023 seen around 10−4 bar for WASP-39 b in Fig. 4. For CH4, the abundance of SO oxidizes the carbon to CO2 via the following mechanism:
 (25)
(25)
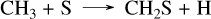 (26)
(26)
 (27)
(27)
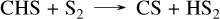 (28)
(28)
 (29)
(29)
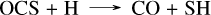 (30)
(30)
 (31)
(31)
Both reactions CHS + S2 → CS + HS2 and CH3 + S → CH2S + H are the limiting steps. The effects of this pathway are more evident when comparing the CH4 abundances from the C/H/O/N kinetic network (dashed lines) with those obtained using the sulfur network (solid lines). This effect is also observed with Tsai 2023, although it does not include the CH2S species, because this oxidation can proceed through the following alternate pathway from CH3 to CS:
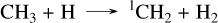 (32)
(32)
 (33)
(33)
 (34)
(34)
 (35)
(35)
The oxidation of CS can then proceed through the same steps.
For WASP-107 b, the situation is very different, as the planet is much cooler. This allows for a huge production of CS2 with our sulfur kinetic network, which predicts CS2 abundances that are over seven orders of magnitude higher than the Tsai 2023 network. Other small differences are observed for SO2 and CH4, for the same reasons discussed above. The abundance of H2S is also affected by variations in the abundance of CS2. The production of C2H2 and C2H4 is strongly favored from the CH4 species through the following pathway:
 (36)
(36)
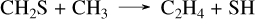 (37)
(37)
 (38)
(38)
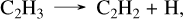 (39)
with the last step being the limiting one. It is also worth noting that the reaction CH2S + CH3 → C2H4 + SH is only approximately 7.3% saturated in this pathway, so it should not be very sensitive to the value of the rate constant; however, its inclusion in the kinetic network is important to allow bypassing the formation of the C2H6 intermediate species, and proceeding directly to the C2H4 product. At 10−3 bar, this represents about 55.7% of C2H2 formation, whereas the standard pathway without sulfur accounts for 14%. A third formation pathway, which contributes 12.1% is noteworthy because it directly involves the species CH3SH,
(39)
with the last step being the limiting one. It is also worth noting that the reaction CH2S + CH3 → C2H4 + SH is only approximately 7.3% saturated in this pathway, so it should not be very sensitive to the value of the rate constant; however, its inclusion in the kinetic network is important to allow bypassing the formation of the C2H6 intermediate species, and proceeding directly to the C2H4 product. At 10−3 bar, this represents about 55.7% of C2H2 formation, whereas the standard pathway without sulfur accounts for 14%. A third formation pathway, which contributes 12.1% is noteworthy because it directly involves the species CH3SH,
 (40)
(40)
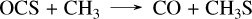 (41)
(41)
 (42)
(42)
 (43)
(43)
 (44)
(44)
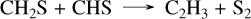 (45)
(45)
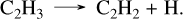 (46)
(46)
The limiting step of this pathway is the reaction CH2S + CHS → C2H3 + S2, which is an analogy made in the network with the addition CH2O + HCO → CH2OCHO;therefore, further work on this reaction is needed to confirm this C2H2 formation mechanism. However, it involves two more reactions that we calculated in Veillet et al. (in rev), which are CH3SH + SH → CH2SH + H2S and CH3S + H2S → CH3SH + SH. These reactions are also key in CH3SH pyrolysis, and our extensive work and validation on experimental data covering these conditions provide high confidence in their rates. For other species such as CH4, the same mechanisms observed for previous planets apply, which are responsible for its oxidation to CO2, the difference between the C/H/O/N and the C/H/O/N/S network in HCN abundance around 10−5 bar, and the difference in NH3 around the same pressure.
The transmission spectra of these planets are shown in Fig. 5. For WASP-39 b, the impact of sulfur coupling on the transmission spectra is negligible. The two features not predicted with the C/H/O/N network are the H2S feature at 4 µm and the SO2 feature at 7.3 µm. Despite the few differences between the networks, both predict a very similar spectrum for this planet.
For WASP-107 b, however, many different features are visible. These features include CS2 at 25 and 11 µm, as well as a different amplitude of about 2000 ppm on the CH4 feature at 3.3 µm, due to the coupling between sulfur and carbon chemistry, which reduces its abundance. The SO2 feature at 7.3 µm also changes the amplitude between models by about 4000 ppm. This indicates that the transit depth is very sensitive to SO2 abundance, as the SO2 differences in abundance are relatively minor between V25 and Tsai 2023. The differences in NH3 abundance also cause the disappearance of the NH3 feature at 20 µm, which is not present in the spectrum corresponding to our sulfur kinetic network but has an amplitude of about 3000 ppm with Tsai 2023. For both planets, we compared our synthetic spectra with observation data, primarily from the JWST. For WASP-39 b, our synthetic spectra appear to correctly reproduce these observational data, but the SO2 feature at 4.1 µm is not observed. This difference is not related to the kinetic network, as we do not reproduce this feature with the Tsai 2023 kinetic network. Since we also used the same planet parameters as in their original work, for which this feature is the main highlight, the absence of this feature is likely linked to differences in the physical modeling of the planet. These differences may arise from both the use of the 1D model FRECKLL and the radiative transfer model TauREx. If due to the 1D model, differences might lie in the simulation parameters, such as the UV flux incident angle or differences in the altitude profile, which is not recomputed from hydrostatic equilibrium while solving for the steady state in the current version of FRECKLL. However, comparison between different radiative transfer models of synthetic spectra generated from the same FRECKLL output shows that some are able to reproduce this SO2 feature at 4.1 µm, strongly suggesting that the differences mainly stem from the radiative transfer modeling. However, for WASP-107 b, using a clear-sky model as presented here cannot reproduce the observed spectrum, which is unsurprising given that silicate clouds have already been proposed to explain observations (Dyrek et al. 2024). We tested simple, optically thick cloud models with TauREx, but identifying suitable cloud pressures and photospheric radii proved too challenging to achieve a satisfactory match between our modeled synthetic spectra and observations. This difficulty also appears related to the puffiness of WASP-107 b, for which the synthetic spectrum was very sensitive to these parameters compared to WASP-39 b. More detailed comparisons with observational data would require careful modeling and a search for an optimal value of these parameters, beyond the scope of this paper, which focuses on the modeling of the chemical kinetics. A more comprehensive parameter study may be conducted in a follow-up paper.
4 Conclusions
In this work, we developed the first sulfur kinetic network extensively validated on combustion and pyrolysis data to model exoplanet disequilibrium chemistry. The network builds on our previous C/H/O/N work (Veillet et al. 2024) and kinetic networks from the combustion literature, which we specifically expanded for CH3SH pyrolysis kinetics with high-level ab initio calculations and analogies to explore a large number of new reactions and yield the most comprehensive sulfur network possible (Veillet et al. in rev). Special emphasis was placed on the coupling of sulfur with all other atoms, including carbon and nitrogen. The coupling between carbon and sulfur has received extensive care and study due to available experimental data for heteroatomic compounds containing sulfur, carbon, and hydrogen together. These kinds of kinetic experimental data are vital for improving our understanding of the reactions involved in these conditions. The species CS2 has received particular attention because it forms during CH3SH pyrolysis. However, existing networks and experiments in the literature mainly focus on CS2 combustion and on pyrolysis, which does not involve hydrogen and therefore cannot be the only source of kinetic data for networks used to model hydrogen-dominated hot exoplanets. We modeled six planets: GJ 436 b, GJ 1214 b, HD 189733 b, HD 209458 b, WASP-39 b, and WASP-107 b, two of which were proven to have at least one sulfur signature species in their atmosphere distinctive of photochemistry, SO2. The simulations highlight a substantial contribution of the CS2 species to the abundance profiles, with abundances as high as 1000 ppm. We computed the corresponding synthetic transmission spectra, which show that this species can contribute to the spectra of the cooler planets. This is in good agreement with recent JWST observations of TOI-270 d (Holmberg & Madhusudhan 2024; Benneke et al. 2024), which indicate potential signatures of CS2 in the spectrum. This finding is clearly relevant for the exoplanet community, as it could potentially be a photochemistry marker for cooler planets, analogous to SO2, and be closely linked to its metallicity. However, some care should be taken, as the full picture of CS2 in exoplanets is far from complete, and further validation should be performed by exploring CS2 oxidation in a hydrogen-heavy medium. This species is not the only one of interest for a reliable and fully coupled C/H/O/N/S network. Other species of interest, such as C2H2 and C2H4, are also affected by the addition of sulfur chemistry, with an increase in abundance of up to two orders of magnitude. Other species, such as CH4, NH3, and HCN, are also found to be impacted by sulfur chemistry, mainly through the central species CH2S, which is key in the coupling of carbon and sulfur chemistry. may contribute significantly to the chemistry through formation from the NS radical, indicating a possible role of nitrogen in the coupling with sulfur chemistry. However, additional work is required to develop a comprehensive network for nitrogen-sulfur chemistry. These results clearly highlight the need for more comprehensive kinetic networks and for extensive validation of our chemistry models in the JWST era. The best tools available for this task are combustion and pyrolysis experiments, which remain the best way to improve the reliability of our photochemistry and thermochemistry models of exoplanet atmospheres.
Data availability
The kinetic network is available on Zenodo (https://doi.org/10.5281/zenodo.17554652), on KIDA (https://kida.astrochem-tools.org/) and on the ANR EXACT website (https://www.anr-exact.cnrs.fr/fr/chemical-schemes/).
Acknowledgements
This project is fund by the ANR project ‘EXACT’ (ANR-21-CE49-0008-01). In addition, O.V. acknowledges funding from the Centre National d’Études Spatiales (CNES). High performance computing resources were provided by IDRIS under the allocation AD010812434R3 made by GENCI and also by the EXPLOR center hosted by the University of Lorraine. This work was partly supported by a Science and Technology Facilities Council Small Award [ST/Y00261X/1].
References
- Abián, M., Cebrián, M., Millera, Á., Bilbao, R., & Alzueta, M. U. 2015, CoFl, 162, 2119 [Google Scholar]
- Al-Refaie, A. F., Changeat, Q., Waldmann, I. P., & Tinetti, G. 2021, ApJ, 917, 37 [NASA ADS] [CrossRef] [Google Scholar]
- Al-Refaie, A. F., Venot, O., Changeat, Q., & Edwards, B. 2024, ApJ, 967, 132 [NASA ADS] [CrossRef] [Google Scholar]
- Alzueta, M. U., Pernía, R., Abián, M., Millera, Á., & Bilbao, R. 2019, CoFl, 203, 23 [Google Scholar]
- Benneke, B., Roy, P.-A., Coulombe, L.-P., et al. 2024, arXiv preprint [arXiv:2403.03325] [Google Scholar]
- Bowman, C. T. & Dodge, L. G. 1977, in Symposium (International) on Combustion, 16 (Elsevier), 971 [Google Scholar]
- Casida, J. E., & Durkin, K. A. 2017, Chem. Res. Toxicol., 30, 94 [Google Scholar]
- Chiang, S.-Y., Lin, I., et al. 2005, JChPh, 122 [Google Scholar]
- Chvatal, V. 1979, Math. Oper. Res., 4, 233 [Google Scholar]
- Colom-Díaz, J., Alzueta, M., Zeng, Z., Altarawneh, M., & Dlugogorski, B. 2021, Fuel, 283, 119258 [Google Scholar]
- Cooper, S. P., Marshall, P., Mathieu, O., et al. 2022, PComI, S1540748922000426 [Google Scholar]
- Cridland, A. J., van Dishoeck, E. F., Alessi, M., & Pudritz, R. E. 2019, A&A, 632, A63 [NASA ADS] [CrossRef] [EDP Sciences] [Google Scholar]
- Drury, C., & Moule, D. 1982, JMoSp, 92, 469 [Google Scholar]
- Dyrek, A., Min, M., Decin, L., et al. 2024, Nature, 625, 51 [Google Scholar]
- Feng, R., Cooper, G., & Brion, C. 1999a, CP, 244, 127 [Google Scholar]
- Feng, R., Cooper, G., Burton, G. R., Brion, C., & Avaldi, L. 1999b, CP, 240, 371 [Google Scholar]
- Feng, R., Cooper, G., Sakai, Y., & Brion, C. 2000, CP, 255, 353 [Google Scholar]
- Finnerty, L., Xuan, J. W., Xin, Y., et al. 2024, AJ, 167, 43 [NASA ADS] [CrossRef] [Google Scholar]
- Fleury, B., Poveda, M., Benilan, Y., et al. 2025, A&A, 693, A82 [NASA ADS] [CrossRef] [EDP Sciences] [Google Scholar]
- Glarborg, P., Halaburt, B., Marshall, P., et al. 2014, JPCA, 118, 6798 [Google Scholar]
- Glarborg, P., Miller, J. A., Ruscic, B., & Klippenstein, S. J. 2018, PECS, 67, 31 [Google Scholar]
- Goodwin, D. G., Moffat, H. K., Schoegl, I., Speth, R. L., & Weber, B. W. 2023, Cantera: An Object-oriented Software Toolkit for Chemical Kinetics, Thermodynamics, and Transport Processes, https://www.cantera.org [Google Scholar]
- Gordon, I., Rothman, L., Hargreaves, R., et al. 2022, JQSRT, 277, 107949 [CrossRef] [Google Scholar]
- Grosch, H., Fateev, A., & Clausen, S. 2015, JQSRT, 154, 28 [NASA ADS] [CrossRef] [Google Scholar]
- He, C., Hörst, S. M., Lewis, N. K., et al. 2020, Nat. Astron., 4, 986 [Google Scholar]
- Heays, A. N., Bosman, A. D., & van Dishoeck, E. F. 2017, A&A, 602, A105 [NASA ADS] [CrossRef] [EDP Sciences] [Google Scholar]
- Higashihara, T., Saito, K., & Yamamura, H. 1976, BCSJa, 49, 965 [Google Scholar]
- Holmberg, M., & Madhusudhan, N. 2024, A&A, 683, L2 [NASA ADS] [CrossRef] [EDP Sciences] [Google Scholar]
- Honorien, J., Sirjean, B., Glaude, P.-A., & Fournet, R. 2023, CoFl, 255, 112913 [Google Scholar]
- Hrodmarsson, H., & Van Dishoeck, E. 2023, A&A, 675, A25 [NASA ADS] [CrossRef] [EDP Sciences] [Google Scholar]
- Hrodmarsson, H. R., Garcia, G. A., Nahon, L., Loison, J.-C., & Gans, B. 2019, PCCP, 21, 25907 [Google Scholar]
- Huebner, W., & Mukherjee, J. 2015, P & SS, 106, 11 [Google Scholar]
- Karan, K., Mehrotra, A. K., & Behie, L. A. 1999, AiChE, 45, 383 [Google Scholar]
- Kempton, E. M.-R., Zhang, M., Bean, J. L., et al. 2023, Nature, 620, 67 [NASA ADS] [CrossRef] [Google Scholar]
- Kreidberg, L., Bean, J. L., Désert, J.-M., et al. 2014, ApJ, 793, L27 [Google Scholar]
- Lira-Barria, A., Rojo, P. M., Mendez, R. A., 2022, A&A, 657, A36 [NASA ADS] [CrossRef] [EDP Sciences] [Google Scholar]
- Lodders, K. 2010, in Astrophysics and Space Science Proceedings, 16, Principles and Perspectives in Cosmochemistry, 379 [Google Scholar]
- Madhusudhan, N., & Seager, S. 2011, ApJ, 729, 41 [Google Scholar]
- Manion, J., Huie, R., Levin, R., et al. 2008, NIST Standard Ref. Database, 17, 20899 [Google Scholar]
- Mathieu, O., Deguillaume, F., & Petersen, E. L. 2014, CoFl, 161, 23 [Google Scholar]
- Mathieu, O., Hargis, J., Camou, A., Mulvihill, C., & Petersen, E. 2015, PComI, 35, 3143 [Google Scholar]
- Molina, L., Lamb, J., & Molina, M. 1981, GeoRL, 8, 1008 [Google Scholar]
- Mollière, P., Wardenier, J., Van Boekel, R., et al. 2019, A&A, 627, A67 [NASA ADS] [CrossRef] [EDP Sciences] [Google Scholar]
- Moses, J. I., Visscher, C., Fortney, J. J., et al. 2011, ApJ, 737, 15 [Google Scholar]
- Moses, J. I., Line, M. R., Visscher, C., et al. 2013, ApJ, 777, 34 [Google Scholar]
- Moses, J., Tsai, S.-M., Fortney, J., et al. 2024, BAAS, 56 [Google Scholar]
- Mukherjee, S., Fortney, J. J., Wogan, N. F., Sing, D. K., & Ohno, K. 2025a, ApJ, 985, 209 [Google Scholar]
- Mukherjee, S., Schlawin, E., Bell, T. J., et al. 2025b, ApJ, 982, L39 [Google Scholar]
- Nixon, M. C., Piette, A. A., Kempton, E. M.-R., et al. 2024, ApJ, 970, L28 [Google Scholar]
- Olschewski, H., Troe, J., & Wagner, H. G. 1994, JPhCh, 98, 12964 [Google Scholar]
- Powell, D., Feinstein, A. D., Lee, E. K., et al. 2024, Nature, 626, 979 [NASA ADS] [CrossRef] [Google Scholar]
- Raj, A., Ibrahim, S., & Jagannath, A. 2020, PECS, 80, 100848 [Google Scholar]
- Roth, P., Löhr, R., & Barner, U. 1982, CoFl, 45, 273 [Google Scholar]
- Sackur, O. 1913, Ann. Phys., 345, 67 [Google Scholar]
- Shiina, H., Oya, M., Yamashita, K., Miyoshi, A., & Matsui, H. 1996, JPhCh, 100, 2136 [Google Scholar]
- Shiina, H., Miyoshi, A., & Matsui, H. 1998, JPCA, 102, 3556 [Google Scholar]
- Sirjean, B., Lizardo-Huerta, J.-C., Verdier, L., Fournet, R., & Glaude, P.-A. 2017, PComI, 36, 499 [Google Scholar]
- Stagni, A., Arunthanayothin, S., Pratali Maffei, L., et al. 2022, ChEnJ, 446, 136723 [Google Scholar]
- Stauffer, E., Dolan, J. A., & Newman, R. 2007, Fire Debris Analysis (Academic Press) [Google Scholar]
- Tennyson, J., & Yurchenko, S. N. 2012, MNRAS, 425, 21 [Google Scholar]
- Tetrode, H. 1912, Ann. Phys., 343, 434 [Google Scholar]
- Tinetti, G., Drossart, P., Eccleston, P., et al. 2018, Exp. Astron., 46, 135 [NASA ADS] [CrossRef] [Google Scholar]
- Tokue, I., Hiraya, A., & Shobatake, K. 1987, CP, 116, 449 [Google Scholar]
- Tsai, S.-M., Lee, E. K., & Pierrehumbert, R. 2022, A&A, 664, A82 [NASA ADS] [CrossRef] [EDP Sciences] [Google Scholar]
- Tsai, S.-M., Lee, E. K., Powell, D., et al. 2023, Nature, 617, 483 [CrossRef] [Google Scholar]
- Vaghjiani, G. L. 1993, JChPh, 99, 5936 [Google Scholar]
- Vandaele, A. C., Hermans, C., & Fally, S. 2009, JQSRT, 110, 2115 [Google Scholar]
- Veillet, R., Venot, O., Citrangolo Destro, F., et al. PCCP, in rev [Google Scholar]
- Veillet, R., Venot, O., Sirjean, B., et al. 2024, A&A, 682, A52 [NASA ADS] [CrossRef] [EDP Sciences] [Google Scholar]
- Venot, O., Hébrard, E., Agúndez, M., et al. 2012, A&A, 546, A43 [NASA ADS] [CrossRef] [EDP Sciences] [Google Scholar]
- Venot, O., Fray, N., Bénilan, Y., et al. 2013, A&A, 551, A131 [NASA ADS] [CrossRef] [EDP Sciences] [Google Scholar]
- Venot, O., Hébrard, E., Agúndez, M., Decin, L., & Bounaceur, R. 2015, A&A, 577, A33 [NASA ADS] [CrossRef] [EDP Sciences] [Google Scholar]
- Venot, O., Bénilan, Y., Fray, N., et al. 2018, A&A, 609, A34 [NASA ADS] [CrossRef] [EDP Sciences] [Google Scholar]
- Venot, O., Bounaceur, R., Dobrijevic, M., et al. 2019, A&A, 624, A58 [NASA ADS] [CrossRef] [EDP Sciences] [Google Scholar]
- Venot, O., Cavalié, T., Bounaceur, R., et al. 2020, A&A, 634, A78 [NASA ADS] [CrossRef] [EDP Sciences] [Google Scholar]
- Wakelam, V., Herbst, E., Loison, J.-C., et al. 2012, ApJS, 199, 21 [Google Scholar]
- Wakeford, H. R., Sing, D. K., Deming, D., et al. 2017, AJ, 155, 29 [NASA ADS] [CrossRef] [Google Scholar]
- Wang, H., Xu, R., Wang, K., et al. 2018, CoFl, 193, 502 [Google Scholar]
- Welbanks, L., Bell, T. J., Beatty, T. G., et al. 2024, Nature, 630, 836 [NASA ADS] [CrossRef] [Google Scholar]
- Woiki, D., & Roth, P. 1994, JPhCh, 98, 12958 [Google Scholar]
- Wu, C. R., & Chen, F. 1998, JQSRT, 60, 17 [Google Scholar]
- Wu, C. R., Chen, F., & Judge, D. 1999, JQSRT, 61, 265 [Google Scholar]
- Xu, J., & Hadjichristidis, N. 2023, Prog. Polym. Sci., 139, 101656 [Google Scholar]
- Xu, Z., Luo, N., Federman, S., et al. 2019, ApJ, 882, 86 [NASA ADS] [CrossRef] [Google Scholar]
- Xue, Q., Bean, J. L., Zhang, M., et al. 2024, ApJ, 963, L5 [NASA ADS] [CrossRef] [Google Scholar]
- Yang, J., & Hu, R. 2024, ApJ, 966, 189 [Google Scholar]
- Zahnle, K., Marley, M. S., Freedman, R. S., Lodders, K., & Fortney, J. 2009, ApJ, 701, L20 [Google Scholar]
- Zeng, Z., Dlugogorski, B. Z., Oluwoye, I., & Altarawneh, M. 2019, CoFl, 210, 413 [Google Scholar]
- Zeng, Z., Dlugogorski, B. Z., Oluwoye, I., & Altarawneh, M. 2021, Fuel, 283, 119257 [Google Scholar]
- Zhou, C. 2009, Kinetic Study of The Oxidation of Hydrogen Sulfide, Ph.D. thesis, The University of Sydney [Google Scholar]
- Zhou, C. R., Sendt, K., & Haynes, B. S. 2013, PComI, 34, 625 [Google Scholar]
In this work, the term “validation” is used in the restricted sense of a systematic confrontation of the kinetic network with the available experimental data, i.e., its ability to reproduce combustion and pyrolysis measurements. It should not be understood as an absolute guarantee of the accuracy of individual rate coefficients, especially for sulfur reactions, where data remain scarce.
Appendix A Photodissociation data
Photodissociation pathways, cross-sections, and quantum yields used in this study for the sulfur species.
Appendix B Transmission spectrum contributions
 |
Fig. B.1 Contribution to the synthetic transmission spectra for all considered absorbing species for the abundances profiles calculated with the present work kinetic network. The plotted quantity is the relative difference to the total transit depth, |
Appendix C Comparisons on CH3SH pyrolysis data
 |
Fig. C.1 Comparison between the kinetic networks from present work (green), Colom-Díaz et al. (2021) (blue) and Tsai et al. (2023) on experimental CH3SH pyrolysis data (N°3 in Table 1) from Alzueta et al. (2019). |
Appendix D Benchmark on the entire combustion and pyrolysis experiments dataset
 |
Fig. D.1 Distribution of each kinetic network prediction errors on the entire experimental dataset, computed as detailed in Veillet et al. (2024). Data is grouped between different conditions of each reference publication in the bataset, based on the fuel studied. The resulting species groups are therefore H2S from Stagni et al. (2022) for conditions 1 to 2 (in cyan), CH3SH from Alzueta et al. (2019) for conditions 3 to 6 (in yellow), CS2 from Zeng et al. (2019) for conditions 7 to 9 (in red), OCS from Zeng et al. (2021) for conditions 10 to 12 (in green), CS2 and OCS with H2O from Abián et al. (2015) for conditions 13 to 15 (in blue) and 16 to 18 (in black). Each condition reference number can be found in Table 1. CH3SH data with water are included in the same group as dry conditions to reduce visual clutter. |
All Tables
Photodissociation pathways, cross-sections, and quantum yields used in this study for the sulfur species.
All Figures
 |
Fig. 1 Comparison between models from the literature for the combustion of 500 ppm of H2S at atmospheric pressure and an air-to-fuel equivalence ratio of four in a flow reactor (no. 1 in Table 1). |
| In the text | |
 |
Fig. 2 Comparison between models from the literature for H2S pyrolysis (500 ppm at atmospheric pressure in a flow reactor, no. 2 in Table 1). |
| In the text | |
 |
Fig. 3 Abundance profiles of GJ 436 b, GJ 1214 b, HD 189733 b, HD 209458 b, WASP-39 b, and WASP-107 b simulated with FRECKLL. Solid lines show results from the present work, while dashed lines show those from Tsai 2023. We omit H2 and He for clarity to focus on the main species. |
| In the text | |
 |
Fig. 4 Abundance profiles of GJ 436 b, GJ 1214 b, HD 189733 b, HD 209458 b, WASP-39 b, and WASP-107 b simulated with FRECKLL. Solid lines show results from the present work, while dashed lines show results from V23. |
| In the text | |
 |
Fig. 5 Synthetic transmission spectra of GJ 436 b, GJ 1214 b, HD 189733 b, HD 209458 b, WASP-39 b, and WASP-107 b simulated with TauREx at a resolution of 50 for the abundance profiles shown in Figs. 3 and 4. |
| In the text | |
 |
Fig. B.1 Contribution to the synthetic transmission spectra for all considered absorbing species for the abundances profiles calculated with the present work kinetic network. The plotted quantity is the relative difference to the total transit depth, |
| In the text | |
 |
Fig. C.1 Comparison between the kinetic networks from present work (green), Colom-Díaz et al. (2021) (blue) and Tsai et al. (2023) on experimental CH3SH pyrolysis data (N°3 in Table 1) from Alzueta et al. (2019). |
| In the text | |
 |
Fig. D.1 Distribution of each kinetic network prediction errors on the entire experimental dataset, computed as detailed in Veillet et al. (2024). Data is grouped between different conditions of each reference publication in the bataset, based on the fuel studied. The resulting species groups are therefore H2S from Stagni et al. (2022) for conditions 1 to 2 (in cyan), CH3SH from Alzueta et al. (2019) for conditions 3 to 6 (in yellow), CS2 from Zeng et al. (2019) for conditions 7 to 9 (in red), OCS from Zeng et al. (2021) for conditions 10 to 12 (in green), CS2 and OCS with H2O from Abián et al. (2015) for conditions 13 to 15 (in blue) and 16 to 18 (in black). Each condition reference number can be found in Table 1. CH3SH data with water are included in the same group as dry conditions to reduce visual clutter. |
| In the text | |
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.